How to Make a Bohr Model: Step-by-Step Guide for Students (2026)
Learn how to draw a Bohr model for any element. Covers electron shell rules, common elements, mistakes to avoid, and when Bohr models break down for heavier atoms.
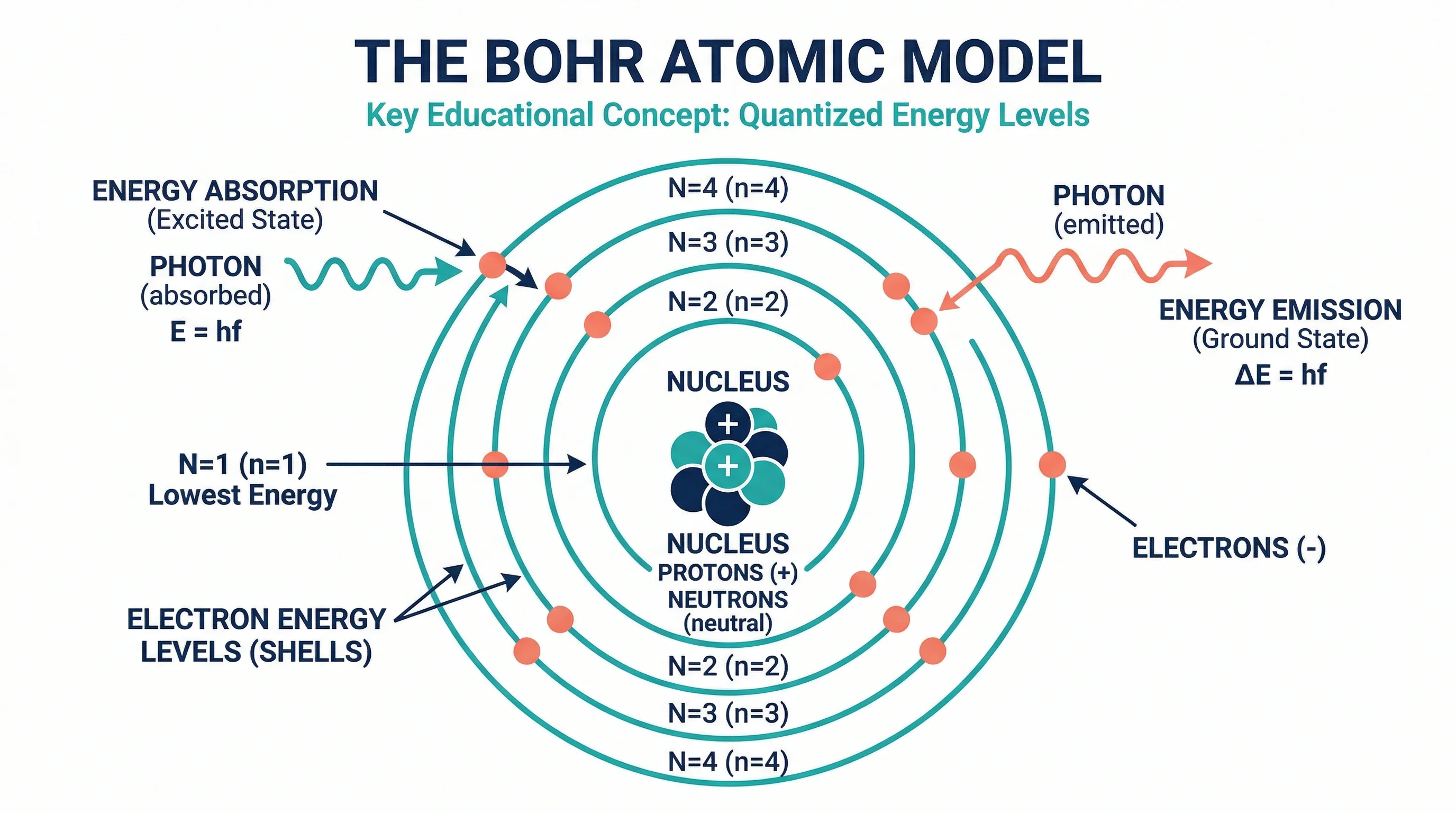
The Bohr model is one of the first atomic models students learn because it makes electron arrangement visual and concrete. Instead of thinking about abstract probability clouds, you draw concentric circles around a nucleus and place electrons on specific shells. That simplicity is why Bohr models remain a standard teaching tool in middle school, high school, and introductory college chemistry courses.
Niels Bohr proposed this model in 1913 to explain hydrogen's emission spectrum. Khan Academy's article on the Bohr model explains how it successfully predicted hydrogen's spectral lines. The model has real limitations — it breaks down for larger atoms and cannot explain molecular bonding well — but for understanding basic atomic structure, it is still one of the clearest starting points.
This guide explains how to draw a Bohr model for any element, what rules govern electron placement, common mistakes, and where the model stops being accurate.
AI Scientific Image Generator
Generate clear scientific diagrams including atomic structures, molecular models, and chemistry visuals from a text description.
Create science diagrams free →What a Bohr Model Shows
A Bohr model represents an atom as:
- a nucleus at the center containing protons and neutrons
- electron shells (energy levels) as concentric circles around the nucleus
- electrons placed on the shells according to specific capacity rules
The model answers the question: "How are the electrons of this atom arranged across energy levels?"
It does not show:
- the actual shape of electron orbitals
- electron spin
- how electrons behave in chemical bonds
- the probabilistic nature of electron location
For those, you need the quantum mechanical model. But for learning the basics of electron configuration, the Bohr model is the right starting tool. A helpful step-by-step tutorial from Middle School Science Blog walks through drawing Bohr diagrams with visual examples.
The Rules for Filling Electron Shells
Before drawing, you need to know how electrons distribute across shells.
Shell capacity rule
Each shell can hold a maximum number of electrons:
| Shell | Name | Maximum Electrons | Formula |
|---|---|---|---|
| 1 | K | 2 | 2(1²) |
| 2 | L | 8 | 2(2²) |
| 3 | M | 18 | 2(3²) |
| 4 | N | 32 | 2(4²) |
The formula is 2n² where n is the shell number.
Filling order
Electrons fill the innermost shell first before moving to the next shell outward. This follows the Aufbau principle at the basic level.
Outermost shell limit (for Bohr models)
For elements in the first three periods of the periodic table, the outermost shell holds a maximum of 8 electrons. This is the octet rule and is the practical limit for most Bohr model exercises.
Important: For elements beyond calcium (Z > 20), the Bohr model becomes increasingly inaccurate because electron filling order no longer follows a simple shell-by-shell pattern.
How to Draw a Bohr Model: Step by Step
Step 1: Find the element's information
Look up the element on the periodic table. You need:
- Atomic number (Z) — this equals the number of protons and, in a neutral atom, the number of electrons
- Mass number (A) — protons + neutrons
Example: Oxygen (O)
- Atomic number: 8
- Mass number: 16
- Protons: 8
- Neutrons: 16 − 8 = 8
- Electrons: 8 (for a neutral atom)
Step 2: Draw the nucleus
Draw a small circle at the center. Inside it, write:
- the number of protons (8p⁺)
- the number of neutrons (8n⁰)
Or simply write the element symbol and mass number: O-16 or 8p, 8n.
Step 3: Draw the electron shells
Draw concentric circles around the nucleus — one for each shell you need.
For oxygen, you need 2 shells (shell 1 and shell 2).
Step 4: Place the electrons
Distribute the 8 electrons following the filling rules:
- Shell 1: 2 electrons (maximum for shell 1)
- Shell 2: 6 electrons (8 − 2 = 6 remaining)
Place the electrons as dots or small circles on the appropriate ring.
Step 5: Verify your count
Total electrons placed should equal the atomic number (for a neutral atom):
- Shell 1: 2
- Shell 2: 6
- Total: 8 ✓
Bohr Models for Common Elements
Here are the electron arrangements for frequently assigned elements:
| Element | Symbol | Atomic # | Shell 1 | Shell 2 | Shell 3 | Shell 4 |
|---|---|---|---|---|---|---|
| Hydrogen | H | 1 | 1 | — | — | — |
| Helium | He | 2 | 2 | — | — | — |
| Carbon | C | 6 | 2 | 4 | — | — |
| Nitrogen | N | 7 | 2 | 5 | — | — |
| Oxygen | O | 8 | 2 | 6 | — | — |
| Neon | Ne | 10 | 2 | 8 | — | — |
| Sodium | Na | 11 | 2 | 8 | 1 | — |
| Chlorine | Cl | 17 | 2 | 8 | 7 | — |
| Argon | Ar | 18 | 2 | 8 | 8 | — |
| Calcium | Ca | 20 | 2 | 8 | 8 | 2 |
Notice how noble gases (He, Ne, Ar) have full outer shells — this pattern is central to understanding chemical reactivity.
Bohr Models for Ions
If the atom has gained or lost electrons, adjust the electron count:
- Positive ions (cations): subtract electrons. Na⁺ has 11 protons but only 10 electrons (2, 8).
- Negative ions (anions): add electrons. Cl⁻ has 17 protons but 18 electrons (2, 8, 8).
The nucleus stays the same — only the electron count changes.
Common Bohr Model Mistakes
Mistake 1: Putting too many electrons on shell 1
Shell 1 can only hold 2 electrons. This is the most common error in student diagrams.
Mistake 2: Forgetting neutrons in the nucleus
The nucleus contains both protons and neutrons. Many students only label the protons.
Mistake 3: Using atomic mass instead of mass number
The atomic mass on the periodic table is a weighted average of isotopes. For a Bohr model, use the mass number of the specific isotope (usually the most common one, which is often close to the rounded atomic mass).
Mistake 4: Not filling inner shells first
Electrons always fill the lowest energy shell first. You cannot put electrons on shell 3 if shell 2 is not full.
Mistake 5: Confusing electrons with protons in the count
The number of electrons equals the atomic number only for neutral atoms. For ions, you must adjust.
Mistake 6: Trying to use Bohr models for transition metals
The Bohr model does not accurately represent elements beyond calcium. Transition metals have complex electron configurations involving d-orbitals that the simple shell model cannot capture.
Where the Bohr Model Breaks Down
The Bohr model is a useful simplification, but it has real limits:
- Multi-electron atoms: The model works best for hydrogen. For larger atoms, electron-electron repulsion and orbital shapes matter.
- Electron orbitals: Real electrons do not orbit in neat circles. They exist in probability clouds (s, p, d, f orbitals) with different shapes.
- Chemical bonding: The Bohr model cannot explain covalent bonding, molecular geometry, or hybridization.
- Transition metals: Elements from scandium (Z=21) onward fill 3d orbitals before completing the 4s shell in some cases, which the simple model cannot represent.
For these topics, the quantum mechanical model and electron configuration notation (1s², 2s², 2p⁶, etc.) become necessary.

Chemistry Structure Generator
Generate molecular structures, Lewis structures, and chemistry diagrams from text descriptions.
Bohr Model vs Lewis Dot Structure
Students sometimes confuse these two:
| Feature | Bohr Model | Lewis Dot Structure |
|---|---|---|
| Shows | All electrons across all shells | Only valence (outer shell) electrons |
| Purpose | Understanding atomic structure | Predicting bonding and molecular shape |
| Format | Concentric circles with dots | Element symbol surrounded by dots |
| Used for | Single atoms | Atoms, molecules, and ions |
Use a Bohr model when you need to show the full electron arrangement. Use a Lewis structure when you need to show bonding potential.
Tips for Drawing Clean Bohr Models
- Use consistent spacing between shells
- Space electrons evenly around each ring (not clumped on one side)
- Label the nucleus clearly with proton and neutron counts
- Keep the diagram simple — avoid decorative elements
- Check your math — total electrons must equal atomic number (for neutral atoms)
- Draw shells you need — do not draw empty shells beyond the outermost occupied one
Frequently Asked Questions
How many electrons can each shell hold in a Bohr model?
Shell 1 holds 2, shell 2 holds 8, shell 3 holds 18, and shell 4 holds 32. The formula is 2n² where n is the shell number. For most classroom Bohr models, the outermost shell is limited to 8 electrons.
How do I make a Bohr model for an ion?
For positive ions (cations), subtract electrons from the neutral atom count. For negative ions (anions), add electrons. The nucleus stays the same — only the electron count changes.
Why does the Bohr model not work for all elements?
The Bohr model assumes electrons orbit in neat circular shells, which is a simplification. For elements beyond calcium, the actual electron filling involves d and f orbitals with complex shapes that the shell model cannot represent.
What is the difference between a Bohr model and a Lewis structure?
A Bohr model shows all electrons across all energy levels for a single atom. A Lewis dot structure shows only valence electrons and is used to predict bonding and molecular shape.
Do I need to draw neutrons in a Bohr model?
Yes. The nucleus should include both protons and neutrons. Label them clearly — for example, 8p and 8n for oxygen.
Can I make a Bohr model for a molecule?
Not really. Bohr models represent individual atoms. For molecules, you need Lewis structures or molecular orbital diagrams that show how atoms share or transfer electrons.
What is the maximum number of elements the Bohr model works for?
The Bohr model is reasonably accurate through calcium (atomic number 20). Beyond that, transition metal electron configurations involve d-orbitals that the simple shell model cannot handle.
Conclusion
Making a Bohr model follows a clear process:
- Find the element's atomic number and mass number
- Draw the nucleus with protons and neutrons
- Draw the electron shells (concentric circles)
- Fill electrons starting from the innermost shell using the 2n² capacity rule
- Verify the total electron count matches the atomic number
The Bohr model is a simplification, but it is a powerful one for learning atomic structure. It makes electron energy levels visual and concrete, which is exactly what students need before moving to the more abstract quantum mechanical model.
For generating science diagrams quickly, try ConceptViz AI Scientific Image Generator or the Chemistry Structure Generator.
Additional Resources:
カテゴリー
もっと読む
ER Diagram Guide: How to Design Entity-Relationship Diagrams Step by Step (2026)
Learn how to draw ER diagrams from scratch with this step-by-step guide. Covers Chen and Crow's Foot notation, design best practices, real examples, free tools, and tips for research databases.

How to Create Science Animations for PowerPoint: Complete Guide for Researchers
Learn to create engaging scientific animations in PowerPoint with zero budget. Includes step-by-step tutorials, free tools comparison, and best practices for molecular biology, chemistry, and physics presentations.
Infographic Design Styles & Trends: Flat, Isometric, Hand-Drawn and Minimalist
Compare infographic design styles and trends: flat, isometric, hand-drawn, minimalist, 3D and editorial layouts, with examples and selection rules.