Lewis Dot Structure Generator
Generate accurate Lewis dot structures for any molecule or ion with AI. Visualize bonding pairs, lone pairs, formal charges, resonance structures, and molecular geometry instantly. Free for students and educators.
Generate Lewis Dot Structure
By using ConceptViz, you agree not to generate or edit adult, sexual, explicit, unsafe, or policy-violating content. See Content Policy.
Free to try ·
Your Lewis dot structure will appear here
Describe a molecule or ion and click Generate
Lewis Dot Structure Examples
Accurate electron dot diagrams for common molecules and ions
Water (H2O) Lewis Structure
Lewis Structure: H2O with bent molecular geometry and lone pairs
Carbon Dioxide (CO2) Lewis Structure
Lewis Structure: CO2 with linear geometry and double bonds
Ammonia (NH3) Lewis Structure
Lewis Structure: NH3 with trigonal pyramidal geometry
Ozone (O3) Lewis Structure
Lewis Structure: O3 resonance structures with formal charges
Sulfate Ion (SO4 2-) Lewis Structure
Lewis Structure: SO4 2- with expanded octet on sulfur
Methane (CH4) Lewis Structure
Lewis Structure: CH4 with tetrahedral geometry and no lone pairs
What is a Lewis Structure?
A Lewis structure (also called a Lewis dot structure or electron dot diagram) is a visual representation of a molecule that shows the arrangement of valence electrons around atoms. Developed by chemist Gilbert N. Lewis in 1916, these diagrams use dots to represent lone pairs and lines to represent bonding pairs of electrons. Lewis structures are fundamental to understanding chemical bonding, molecular geometry, and reactivity. Our free Lewis Dot Structure Generator uses AI to create accurate electron dot diagrams for any molecule or ion, helping you visualize covalent bonds, lone pairs, formal charges, and resonance structures instantly.
Rules for Drawing Lewis Structures
- Count Valence Electrons: Sum up all valence electrons from each atom in the molecule, adjusting for ionic charges
- Identify the Central Atom: The least electronegative atom (not hydrogen) is typically the central atom
- Draw Single Bonds First: Connect each outer atom to the central atom with a single bond (2 electrons per bond)
- Distribute Remaining Electrons: Place remaining electrons as lone pairs on outer atoms to satisfy the octet rule
- Check the Octet Rule: Ensure each atom has 8 electrons (2 for hydrogen) by forming multiple bonds if needed
- Calculate Formal Charges: Verify that formal charges are minimized and negative charges are on more electronegative atoms
Common Lewis Structures for Students
Chemistry students frequently need Lewis structures for common molecules like water (H2O), carbon dioxide (CO2), ammonia (NH3), methane (CH4), oxygen (O2), nitrogen (N2), and ozone (O3). More advanced structures include polyatomic ions such as sulfate (SO4 2-), nitrate (NO3-), and phosphate (PO4 3-), as well as molecules with expanded octets like sulfur hexafluoride (SF6) and phosphorus pentachloride (PCl5). Our generator handles all of these, including resonance structures, formal charge calculations, and exceptions to the octet rule.
Lewis Structures in Education
Lewis dot structures are a cornerstone of chemistry education from high school through graduate school. They serve as the foundation for understanding VSEPR theory (Valence Shell Electron Pair Repulsion), which predicts molecular geometry. By drawing Lewis structures, students learn to predict bond angles, molecular polarity, hybridization, and chemical reactivity. Our Lewis Dot Structure Generator is designed to help students verify their hand-drawn structures, study for exams, and create professional diagrams for lab reports and presentations.
Frequently Asked Questions
More Chemistry Tools
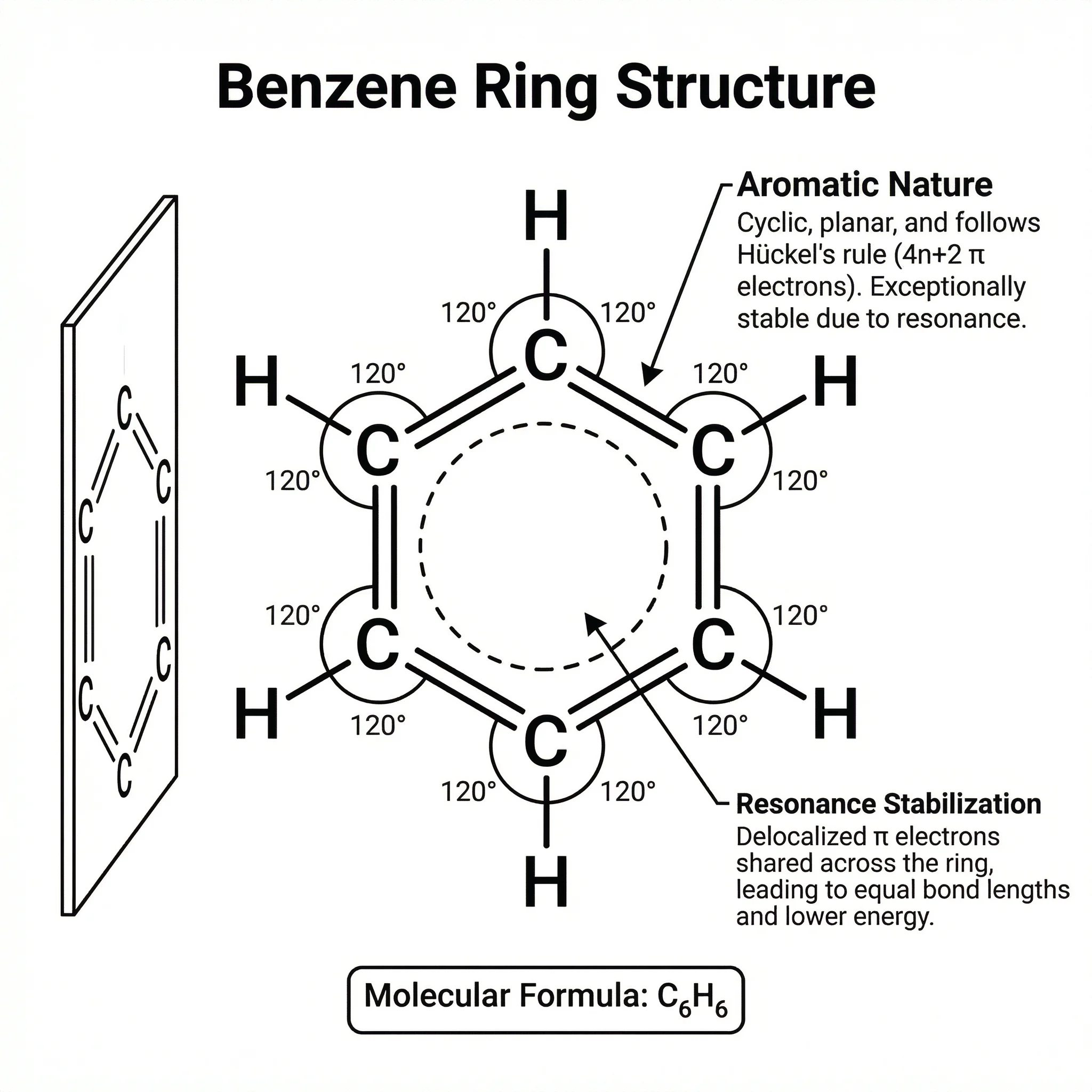 Chemistry
ChemistryChemistry Structure Generator
Create molecular structures, reaction mechanisms, and chemical diagrams with AI.
 Chemistry
ChemistryChemistry Poster Maker
Create professional chemistry posters including periodic tables and reaction diagrams.
 Research
ResearchAI Scientific Image Generator
Generate professional scientific images and illustrations for research papers.